Get premium membership
Get premium membership and access revision papers with marking schemes, video lessons and live classes.
In an experiment determine the heat of combustion of methanol, #CH_3OH#, a student used a set up like the one shown in the diagram below.
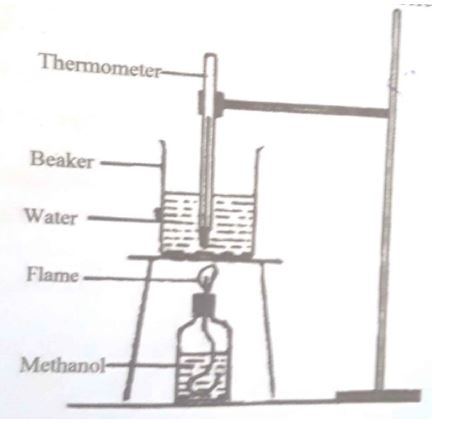
Volume of water=#500cm^3#
Final temperature of water=#27.0^0C#
Initial temperature of water=#20.0^0C#
Final mass of lamp+methanol=22.11g
Initial mass of lamp+methanol=22.98g
Density of water=1.0g#cm^3#
Specific heat capacity of water=4.2J/g/K
(a)Write an equation for the combustion of methanol
(b)Calculate: (i)The number of moles of methanol used in this experiment (C=12;O=16;H=1)
(ii) The heat of combustion of methanol.
(iii) The heat of combustion per mole of methanol
(c ) Explain why the value of the molar heat of combustion for methanol obtained in this experiment is different from the theoretical value.
(d) On the axis below draw an energy level diagram for the combustion of methanol.