 Get premium membership
Get premium membership and access revision papers with marking schemes, video lessons and live classes.
In order to determine the molar heat of neutralization of sodium hydroxide, 100#cm^3# of 1M sodium hydroxide and 100#cm^3# of 1M hydrochloric acid both at the same initial temperature were mixed and stirred continuously with a thermometer. The temperature of the resulting solution was recorded after
every 30 seconds until the highest temperature of the solution was attained. There after, the temperature of the solution was recorded for a further two minutes.
(a)(i)Why was it necessary to stir the mixture of the two solutions?
(ii) Write an ionic equation for the reaction which took place.
(iii) The sketch below was obtained when the temperature of the mixture were plotted against time. Study it and answer the questions that follow.
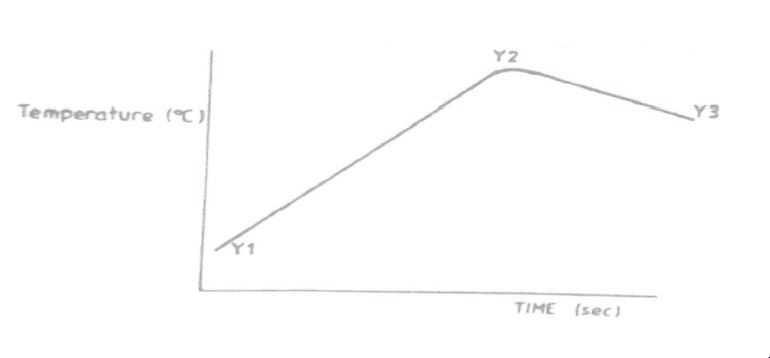
I.What is the significance of point Y2?
II.Explain why there is a temperature change between points.
Y1 and Y2