 Get premium membership
Get premium membership and access revision papers with marking schemes, video lessons and live classes.
(a) The diagram below shows some processes that take place during the industrial manufacture of sulphuric (VI) acid.
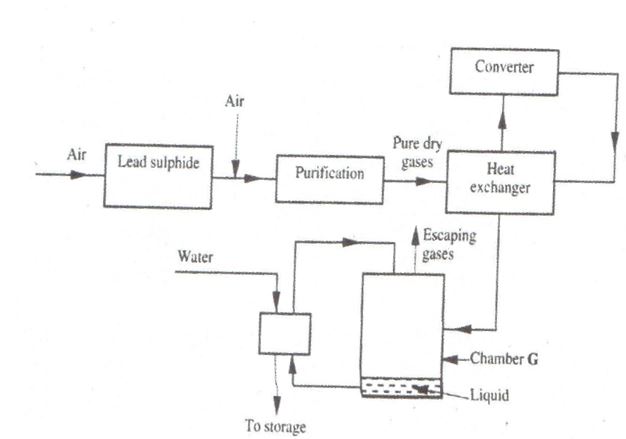
(i) Write the equation for the reaction in which sulphur (IV) oxide gas is produced
(ii) Why is it necessary to keep the gases pure and dry?
(iii) Describe the process that takes place in chamber G
(iv) Name the gases that escape into the environment
(v) State and explain the harmful effect on the environment of one of the gases named (iv) above.
(vi) Give one reason why it is necessary to use a pressure of 2-3 atmospheres and not more.
(b) (i) Complete the table below to show the observation made when concentrated sulphuric (VI) acid is added to the substances shown.
(ii) Give reasons for the observation made using I iron fillings
II crystals of sugar
(c) Name one fertilizer made from sulphuric (VI) acid.