 Get premium membership
Get premium membership and access revision papers with marking schemes, video lessons and live classes.
The set up below was used to prepare a gas and study some of its properties. Study it and answer the questions
that follow:
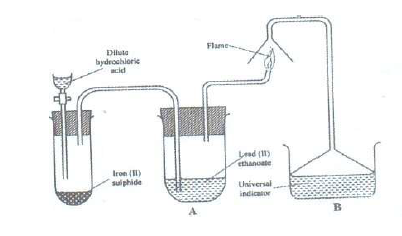
(a) State and explain the observations made in the:
(i) Tube labeled A;
(ii) Beaker labeled B
(b) State one precaution that should be taken when carrying out this experiment.