 Get premium membership
Get premium membership and access revision papers with marking schemes, video lessons and live classes.
The set-up below was used to prepare anhydrous chlorides of a number of elements in a laboratory where no fume cupboard was available. The chlorides were to be collected in flask 1.
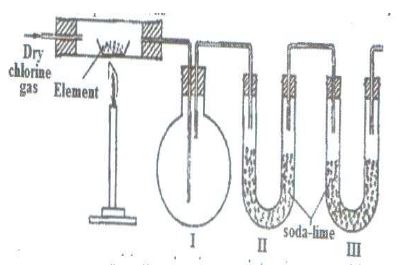
The following table shows the melting and boiling points of the chlorides that were prepared.
(a) Explain why it is necessary to pass dry chlorine through the apparatus before heating each element.
(b) Give two reasons why tubes II and III were filled with soda lime (solid mixture of sodium hydroxide and calcium hydroxide).
(c) Explain why it would not be possible to collect any sodium chloride in flask 1.
(d) Name one other substance that can be used in tubes II and III.
(e) Write an equation for the reaction that forms phosphorous (III) chloride.
(f) Describe how you would separate a mixture of sodium chloride and aluminium chloride.