 Get premium membership
Get premium membership and access revision papers with marking schemes, video lessons and live classes.
Figure 3 is a flow chart that shows the process that occurs in the manufacture of nitric(v) acid, it produces a nitrogenous fertilizer
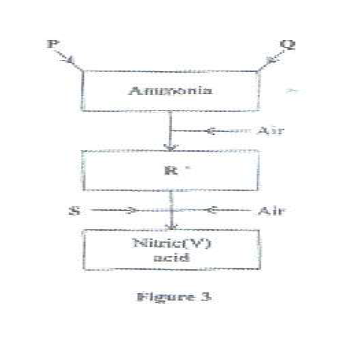
(a) Name substance P, Q, R and S. P
(b) To obtain substance R, ammonia is heated at 900°C in the presence of air and a catalyst.The product is then cooled in air.
(i) Name the catalyst for the reaction.
(ii) Write the equations for the two reactions described in (b).
(iii) Other than nitric(V) acid, name another product that is formed.
(c) When ammonia is reacted with nitric(V) acid, it produces a nitrogenous fertiliser. State two problems associated with the use of nitrogenous fertilisers.