 Get premium membership
Get premium membership and access revision papers with marking schemes, video lessons and live classes.
(a) The diagram in Figure 4 was used to prepare hydrogen chloride gas which was passed over heated iron powder.
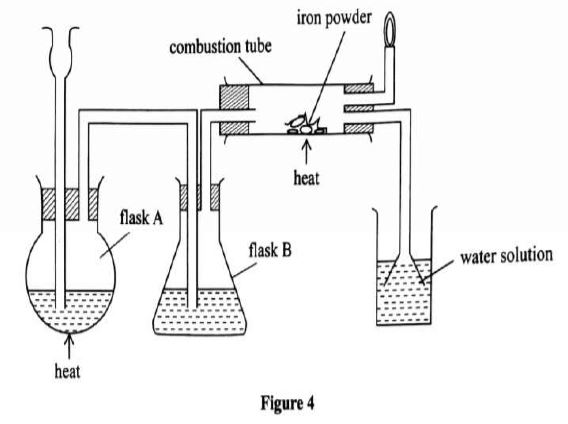
(i) Give a pair of reagents that will produce hydrogen chloride gas in flask A.
(ii) Name the substance in flask B.
(iii) State the observation made in the combustion tube.
(iv) Write an equation for the reaction in the combustion tube.
(iv) Describe a chemical test for hydrogen chloride gas.
(b) (i) Identify the gas that bums at the jet.
(ii) Explain why the gas in (b) (i) is burned.
(c) Give reasons why excess hydrogen chloride gas is dissolved using the funnel arrangement.
(d) State what will be observed when the reaction in the combustion tube is complete.