 Get premium membership
Get premium membership and access revision papers with marking schemes, video lessons and live classes.
(a) Two reagents that can be used to prepare chlorine gas are manganese (IV) oxide and concentrated hydrochloric acid.
(i) Write an equation for the reaction
(ii) Give the formula of another reagent that can be reacted with concentrated hydrochloric acid to produce chlorine gas.
(iii) Describe how chlorine gas could be dried in the laboratory.
(b) In an experiment, dry chlorine gas was reacted with aluminium as shown in figure 1
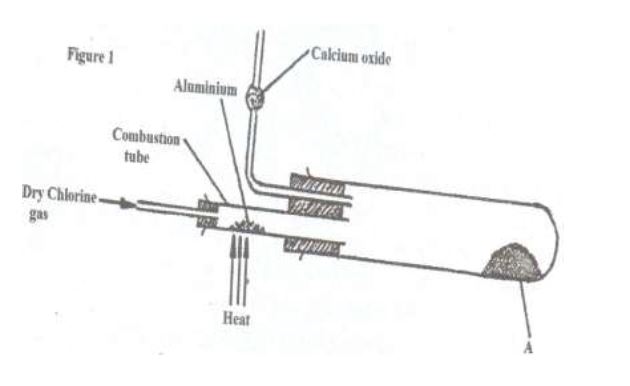
(i) Name substance A
(ii) Write an equation for the reaction that took place in the combustion tube .
(iii) 0.84g of aluminium reacted completely with chlorine gas . calculate the volume of chlorine gas used (molar gas volume is 24#dm^3#, Al=27)