 Get premium membership
Get premium membership and access revision papers with marking schemes, video lessons and live classes.
(a) State two factors that affect the boiling point of a liquid ( 2 mks) (b) 100g of a liquid at a temperature of 100C is poured into a well lagged calorimeter . An electric heater rated 50W is used to heat the liquid. The graph in figure 8 shows the variation of the temperature of the liquid with time. Figure 8 (i) From the graph, determine the boiling point of the liquid ( 1 mk)
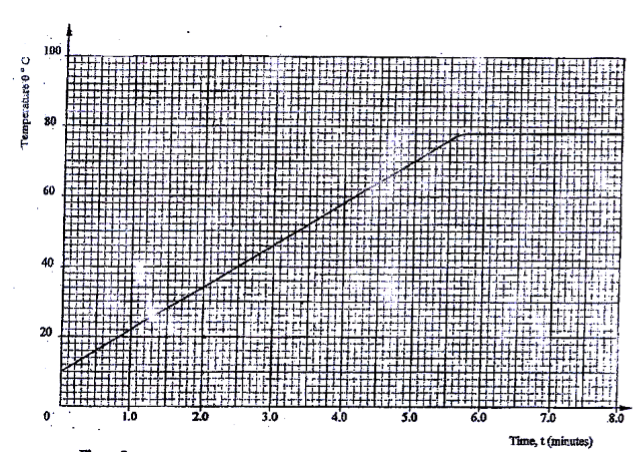
(ii) (I) Determine the heat given out the by the heater between the times t= 0.5 minutes and t= 5.0 minutes ( 2 mks) (II) From the graph determine the temperature change between
the times t = 0.5 minutes and t= 5.0 minutes ( 1 mk) (III) Hence determine the specific heat capacity of the liquid ( 2 mks) (iii) 1.8 g of vapour was collected from above the liquid between the times t= 6.8 minutes and t= 7.3 minutes. Determine the specific latent heat of vaporization of the liquid ( 4 mks)