 Get premium membership
Get premium membership and access revision papers with marking schemes, video lessons and live classes.
Form 2 Chemistry End of Term 3 Examination 2021
Class: Form 2
Subject: Chemistry
Level: High School
Exam Category: Form 2 End Term 3 Exams
Document Type: Pdf
Views: 1469
Downloads: 74
Exam Summary
TUTORKE EXAMS
NAME……………………………………………… ADM NO……………… CLASS……………………….
CHEMISTRY.
FORM 2.
TIME: 2 HOURS.
END OF TERM THREE EXAMINATION 2022
INSTRUCTION TO CANDIDATES
Answer all the questions in the spaces provided.
1. The diagram below shows parts of the Bunsen burner.
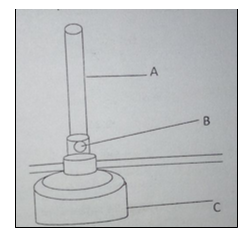
(b) Give one use of the part labelled B (1 mark).
2. Name two apparatus that are used to measure exact volumes (2 marks).
3. The diagram below represents a method of separation used to separate two liquids, A and B,
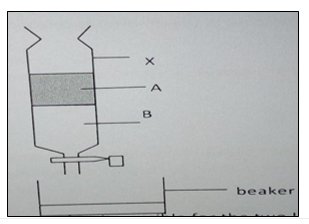
Use it to answer the questions that follow.
(a) Name the method of separation shown above (1 mark).
(b) Name two properties that makes it possible for the two liquids to be separated (2 marks).
(c) Give one alternative method that may be used to separate the two liquids (1 mark).
4. In an experiment, two pieces of iron sheet were wrapped in each case with zinc and copper metal sheets as shown below. They were left in the open for some months
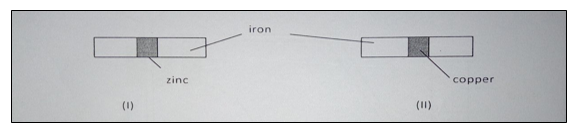
(a) State and explain the observations made in the experiment I and II (3 marks).
I
II
5. Given zinc oxide, dilute nitric (v) acid and sodium carbonate solution. Briefly describe how
you can prepare zinc carbonate (3 marks).
6. State two solids that may be heated to obtain oxygen gas as the only gas (2 marks).
7. Study the information given below and answer the questions that follow.
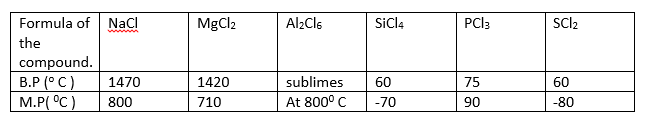
(a) Give two chlorides that are liquid at room temperature. Give a reason for the answer
(2 marks)
(b) Which two chlorides would remain in liquid state for the highest temperature range. Explain. (2 marks).
(c) State one use of NaCl (1 mark).
8. Copper (II) oxide reacts with zinc metal as shown below.
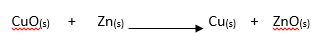
Identify the substance that has been:
(i) Reduced (1 mark)
(ii) Oxidised (1 mark)
9. An element B burns in air to form an oxide of B which dissolve in water to form a solution
that turns blue litmus paper red. (B is not the actual symbol of the element).
Is B a metal or a non-metal? Give a reason for your answer (2 marks).
10. Study the diagram below and answer the questions that follow.
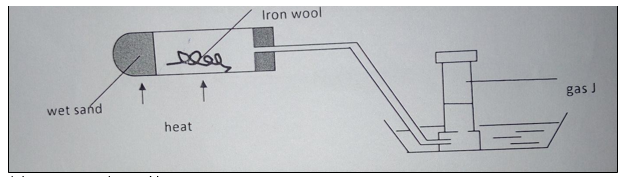
(a) Name gas J (1 mark).
(b) Explain why it is important to heat the wet sand before heating the iron wool (1 mark).
(c) State one observation made in the combustion tube as heating is carried out (1 mark).
11. State two differences between permanent and temporary changes (2 marks).
12. The table below give some properties of substances I, J and K. Study it and answer the questions that follow.
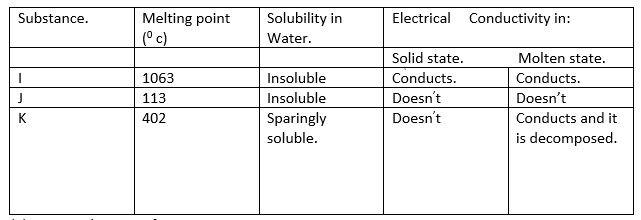
(a) Suggest the type of structure in:
I (1 mark).
K (1 mark).
(b) Explain why molten K is decomposed by current but I is not decomposed (1 mark).
13. Solution R,S and T have pH values shown in the table below.
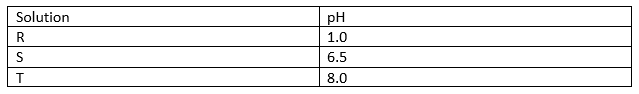
(a) What do you deduce about the nature of solution R? (1 mark).
(b) Identify two solutions that will react to form a neutral solution (1 mark).
14. State and explain the changes in mass that occur when zinc metal is heated in an open
crucible (2 marks).
15. The Ph of the soil sample was found to be 6.0. An agricultural officer recommended the
addition of lime(calcium oxide). State two functions of lime in the soil (2 marks).
16. (a) Using a dot(.) and cross(x) to represent the outer most electrons, draw diagrams to show
the bonding in magnesium sulphide (2 marks). (Mg=12,S=16).
(b) State the structure of the above compound (1 mark).
(c) Give two properties of substances with the above structure (2 marks).
17. In an experiment a test tube full of chlorine water was inverted in chlorine water as shown in the diagram and the set up left in the sunlight for one day.
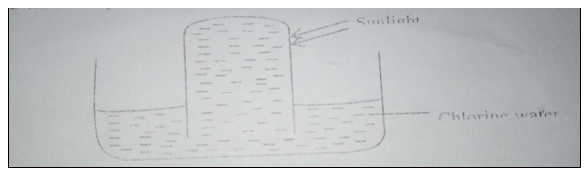
After one day a gas was found to have collected in the test tube.
(a) Identify the gas (1 mark).
(b) State the observations made when a blue litmus paper is dipped in chlorine water (1 mark).
18. The elements shown in the table below (not actual symbols) belong to a certain family of metals in the periodic table. Study the information and answer the questions that follow.
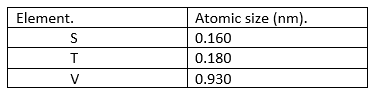
(i) Define the term ionization energy (1 mark).
(ii) Which element is likely to have the highest ionization energy. Explain (2 marks).
19. State two applications of solvent extraction (2 marks).
20. The diagram below represents the structure of aluminum chloride.
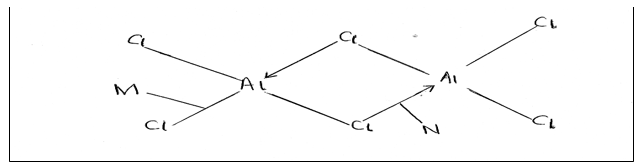
a) Identify the bonds labeled M and N (2 marks).
M
N
(b) What is the difference between bonds M and N (1 mark).
21. From the following list of compound; Zinc oxide, Potassium carbonate, solid carbon (Iv)
oxide, nitric acid, iron(III)chloride, sodium chloride.
(i) Identify two substances that sublime (1 mark).
(ii) Identify two substances that react to form salt and water only (1 mark).
22. The figure below is a set up used to investigate the reaction of calcium with water.
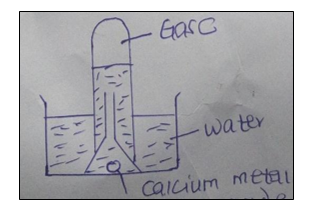
(a) State the observation made in the water (1 mark).
(b) Identify gas C (1 marks).
(c) State one laboratory application of the solution formed in the reaction (1 mark).
23.(i) State two types of salts (1 mark).
(ii) Name the following processes;
(a) When anhydrous calcium chloride is left in an open beaker overnight a solution was
formed (1 mark).
(b) When sodium carbonate decahydrate crystals are left in an open beaker for some
days it turned into a powder (1 mark).
24. The following diagrams show the structures of two allotropes of carbon. Study them and answer the questions that follow.
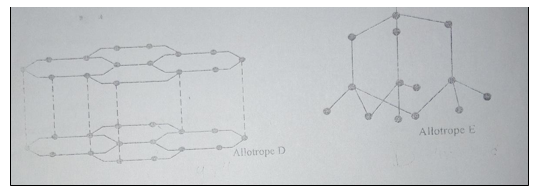
(a) Name the allotrope D and E (2 marks).
D
E
(b) Which allotrope does not conduct electricity? Explain (2 marks).
25. Iron(III)chloride can be prepared in the laboratory by passing dry chlorine gas over hot steel
wool.
(a) Name the above method of preparing salts (1 mark).
(b) Why should we prepare the salt in a dry environment? (1 mark)
(c) A solution of iron (III) chloride in water changes a blue litmus paper to red. Explain.
(1mark).
26. Metal S removes oxygen combined with P. Q reacts with an oxide of R but not with an oxide
of P. P reacts with cold water but Q does not.
(a). Which is the most reactive metal? (1 mark).
(b) Which is the least reactive metal? (1 mark).
(c). Arrange the metals in order of reactivity starting with the most reactive to the least
reactive (1 mark).
27. The figure represents a set up of apparatus that was used to demonstrate the existence of a component of air.
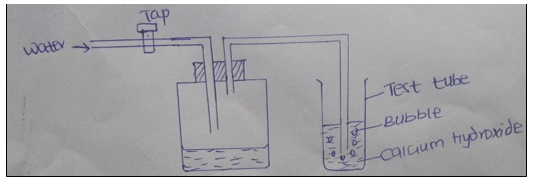
(i) What is the purpose of water from the tap? (1mark).
(ii) Other than bubbles, give one other observation made in the test tube (1 mark).
(iii) Name a gas that was not absorbed by the calcium hydroxide. (1mk)
28. The grid below is part of the periodic table. Study it and answer the questions that follow. The letters do not represent the actual symbols of the elements.
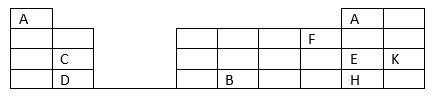
(a) Element A fits in two groups. Explain (2 marks).
(b) i) Name two elements that can form ions with a charge of -1. Explain your answer (2 marks).
ii) What type of structure would the oxide of B have? (1 mark).
(c) How does the reactivity of H compare with that of E? Explain. (2 marks)
(d) What name is given to the group of elements to which C and D belong? (1 mark).
(e) Write the formula of the compound formed when elements C and F react (1 mark).
(f) Draw dot (.) and cross (x) diagram to represent the compound formed in (e) above (2 marks).
29. A student set up the arrangement below to prepare and collect dry hydrogen gas.
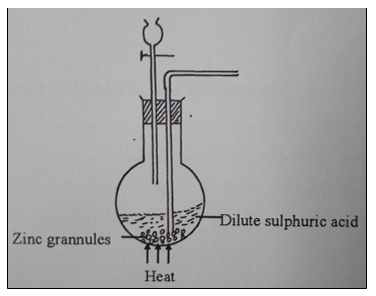
(a) State the catalyst that is used in preparation of hydrogen gas (1 mark).
(b) Identify two errors from the section of the arrangement shown above (2 marks).
(c) Complete the diagram to show how dry hydrogen gas can be collected (2 marks).
(d) Explain the effect of hydrogen gas on a wet red litmus paper (1 mark).
(e) Write a balanced chemical equation for the reaction that takes place when hydrogen gas is burnt in air. (1 mark).
(f) State one property of hydrogen gas that makes it to be used in balloons (1 mark).
30. Study the flow chart below and answer the questions that follow.
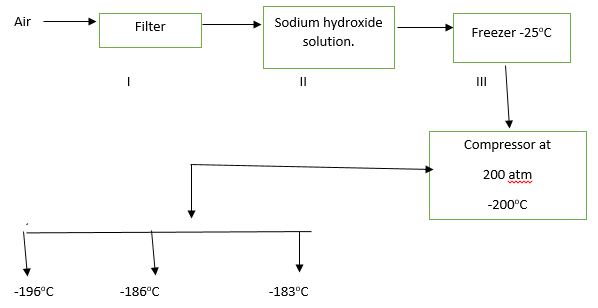
(a) Name the substances removed in steps I,II and III (3 marks).
I
II
III
(b) Name the gases obtained with respect to their boiling points (3 marks).
-1960C
-1860C
-1830C
More Examination Papers