 Get premium membership
Get premium membership and access revision papers with marking schemes, video lessons and live classes.
Form 4 Chemistry Paper 2 End of Term 2 Exams 2021
Class: Form 4
Subject: Chemistry
Level: High School
Exam Category: Form 4 End Term 2 Exams
Document Type: Pdf
Views: 816
Downloads: 40
Exam Summary
233/2
CHEMISTRY
PAPER 2
END OF TERM 2 EXAM 2021
1. The grid below shows a section of the periodic table, the letters are not the actual chemical symbol.
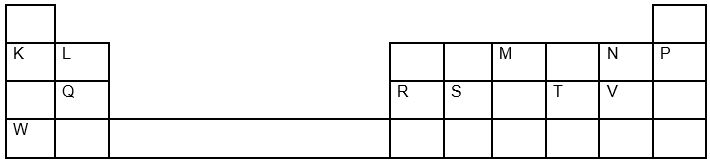
a) Name the family into which element P belongs to ( 1mk)
b) Which two elements forms the most soluble carbonates (2mks )
c) With a reason, identify elements in period 3 with the largest atomic radius (2mks )
d) Write the formula of the compound formed between Q and M (1mk )
e) State two uses of element R and for each use , state property of element R that makes it possible for the use
Use ( 1mk)
Property (1mk)
Use (1mk)
Property (1mk)
f) Using dots and cross ,show bonding in the compound formed between R and oxygen (2mks )
g) In terms of structure and bonding explain why the oxides of element T has relatively low boiling points (2mks)
2. (a) name the following compounds (3mks)
(i) #CH_3CH_2CH_2COOH #
(ii) 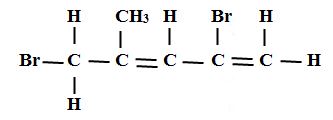
(iii) #CH_3CH_2OOCCH_2CH_3#
b) Two types of detergents P and Q can be represented as
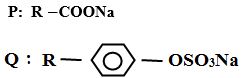
(i) Identify each type of the detergent (2mks)
(ii) Which of the two detergents is the best to use with hard water? Give a reason (2mks)
(iii) State one advantage of detergent P (1mk)
(iv) State one disadvantage of detergent Q (1mk)
(c) An hydrocarbon can be represented as follows
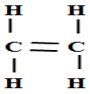
(i) Identify the hydrocarbon (1mk)
(ii) Name two reagents that can reacted together to generate the hydrocarbon (2mks)
3. (a) Name two apparatuses that can be used for determining mass in a laboratory (2mks)
(b) One of the flames produced by Bunsen burner is the luminous flame
i) Explain why this flame is very bright (1mk )
ii) State two disadvantages of the luminous flame (2mks)
(c) Air is usually one of the substances that is considered as a mixture
(i) Identify the two most abundant component of air (2mks )
(ii) Give two reasons why the air is considered as a mixture (2mks)
(iii) One of the components of air is carbon (iv) oxide. Describe an experiment that can be used to prove the presence of carbon (iv) oxide in the air (2mks)
4. (a) The diagram below shows the process used to obtain Sulphur from underground deposits
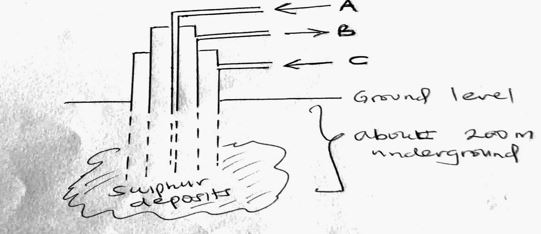
i) Name the above process used to obtain sulphur from the underground deposits (1mk)
ii) Name the substance passed through pipe
A (1mk)
B (1mk)
iii) State two properties of Sulphur that makes it possible to extract using the above process (2mks)
b) The diagram below shows the contact process used in the manufacture of concentrated sulphuric(vi) acid
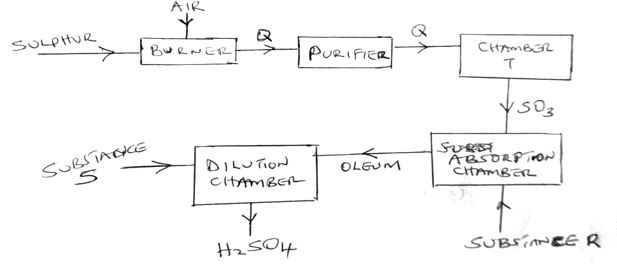
i) Identify the following:
a) Substance Q formed in the burner (1mk)
b) Chamber T (1mk)
c) Substance R (1mk)
d) Substance S (1mk)
ii) Write the chemical equation occurring in the dilution chamber (1mk)
iii) Why is it necessary to pass substance Q though a purifier (1mk)
iv) State one use of sulphuric (VI) acid (1mk)
5. (a) Calamine is one of the ores from which zinc can be extracted from
(i) Name any other ore from which zinc can be extracted from (1mk)
(ii) The calamine is usually decomposed by heating to obtain substance M as shown below

Identify substance M (1mk)
(iii) Identify two methods that can be used to obtain zinc from substance M (2mks)
(b) During the extraction of zinc, name two gases likely to emitted into the air and that are likely to cause pollution (2mk)
(c) State one likely pollution effects of each of the gases you have mentioned in (a) above (2mks)
(d) State one possible use of zinc metal (1mk)
6. (a) define the term electrolysis (1mk)
(b) State two functions of a salt bridge during electrolysis (2mks)
(c) The reduction potential of elements K, L, M, and P are as given below.

Which letter represents the, strongest reducing agent? give a reason (2mks)
(ii) Which two letters represent elements whose half cells would form an electrochemical cell with the largest e.m.f? (1mk)
(iii) Calculate the e.m.f of the cell formed in (ii) above (2mks)
(d) During the electrolysis of a molten chloride of metal Q, a current of 0.25A was passed though the molten chloride for 2 hours and 10minutes. Given that 0.9grams of metal Q were deposited at the cathode.
(i) Calculate the quantity of electricity passed (1mk)
(ii) Charge carried by the ions of metal Q given that R.A.M of metal Q is 84 (3mks)
7 (a) starting with magnesium oxide, describe how you can obtain a dry sample of magnesium Carbonate (3mks)
(b) (i) Give one example of an acid salt ( 1mk)
(ii) When sodium nitrate was heated a solid A and gas B were produced identify solid A and gas B (2mks)
(iii) State two uses of gas B produced in (ii) above (2mks)
(c) State two factors that should be considered when choosing a fuel (2mks)
More Examination Papers